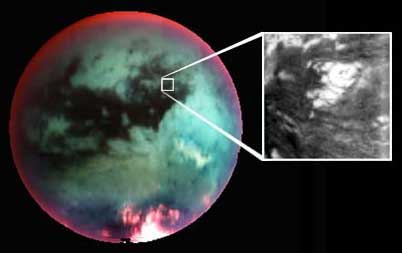
Cassini-Huygens spacecraft and Titan (c) NASA
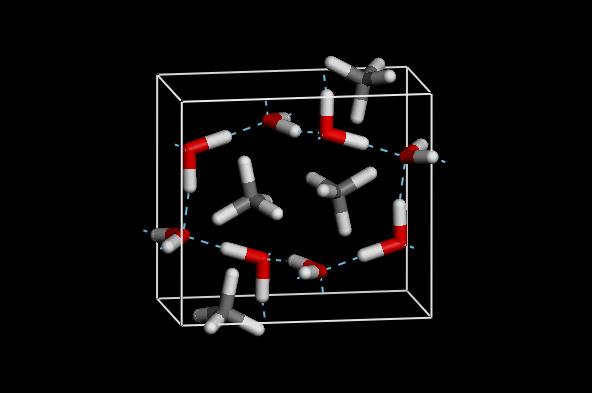
High Pressure Phase of Mathane Hydrate: MH-III (c) T.Iitaka
| Japanese | English |
News | Download |
Research | Study | Links | Japan | Who am I | RIKEN | Clock |
 Cassini-Huygens spacecraft and Titan (c) NASA |
 High Pressure Phase of Mathane Hydrate: MH-III (c) T.Iitaka |
Methane hydrate (MH), known as Burning
Ice, is a special class of ice that contains methane molecules
in cages or networks of hydrogen bonded water molecules. Low pressure
phase of methane hydrate (MH-I) forms sI structure of cages. MH-I,
abundant in the deep ocean, has been attracting attention of the
industry as a key material of new energy resource, whose amount is
estimated twice as much as the total fossil fuel reserve.
References